Antibody-drug conjugates (ADCs) represent a novel strategy in targeted cancer therapy. Combining the strong, lethal effects of chemotherapeutic medicines with the precise targeting potential of monoclonal antibodies. Applications of glycoscience reagents, which are essential for improving the effectiveness, stability, and specificity of these intricate molecular structures, are at the core of this novel therapeutic approach.
Glycoscience reagents provide unique molecular tools that address several critical challenges in antibody-drug conjugation.
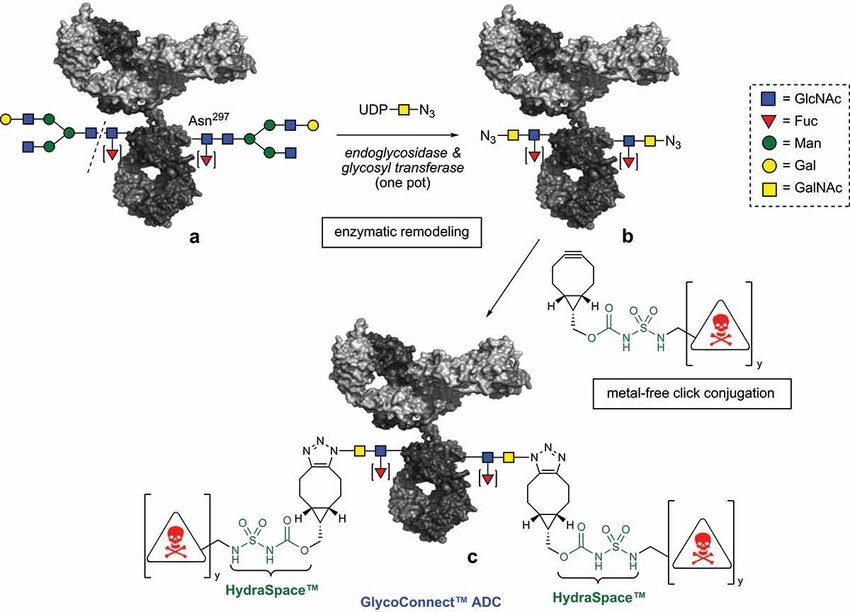
Picture From Reference Number 7
Precise Conjugation Chemistry
Reagents based on glycosylation allow for more precise and regulated drug attachment to antibodies. Conventional conjugation techniques frequently produce diverse mixes with fluctuating drug-to-antibody ratios (DAR). Engineered glycan structures are introduced using applications of glycoscience approaches, which act as precise chemical handles for drug linkage.
Enhanced Stability and Pharmacokinetics
ADC stability can be greatly increased by glycosylation changes, which also minimize systemic toxicity and premature drug release. Researchers can maximize the pharmacokinetic characteristics of these medicinal compounds by deliberately altering glycan structures.
Applications
- Site-Specific Conjugation by selectively altering the glycans on mAbs, Applications of glycoscience reagents allow to produce homogeneous ADCs. Enzymes like galactosyltransferases or sialyltransferases are used in techniques like enzymatic glycoengineering to add functional groups for conjugation.
- Reduced Immunogenicity as the engineered glycans lower the possibility of immunogenic reactions, ADCs are safer to utilize in clinical settings. This is particularly important when patients are receiving repeated doses.
- Improved pharmacokinetic characteristics of ADCs by modifications employing glycoscience reagents. For example, sialylation lengthens the half-life of serum, enabling longer therapeutic action and fewer doses.
References
- Zhu, Z., Ramakrishnan, B., Li, J., Wang, Y., Feng, Y., Prabakaran, P., … & Dimitrov, D. S. (2014, September). Site-specific antibody-drug conjugation through an engineered glycotransferase and a chemically reactive sugar. In MAbs (Vol. 6, No. 5, pp. 1190-1200). Taylor & Francis.
- Shi, W., Li, W., Zhang, J., Li, T., Song, Y., Zeng, Y., … & Huang, W. (2022). One-step synthesis of site-specific antibody–drug conjugates by reprograming IgG glycoengineering with LacNAc-based substrates. Acta Pharmaceutica Sinica B, 12(5), 2417-2428.
- Wijdeven, M. A., van Geel, R., Hoogenboom, J. H., Verkade, J. M., Janssen, B. M., Hurkmans, I., … & van Delft, F. L. (2022, December). Enzymatic glycan remodeling–metal free click (GlycoConnect™) provides homogenous antibody-drug conjugates with improved stability and therapeutic index without sequence engineering. In MAbs (Vol. 14, No. 1, p. 2078466). Taylor & Francis.
- Zhou, Q., Stefano, J. E., Manning, C., Kyazike, J., Chen, B., Gianolio, D. A., … & Pan, C. Q. (2014). Site-specific antibody–drug conjugation through glycoengineering. Bioconjugate chemistry, 25(3), 510-520.
- Spiro, R. G. (2002). Protein glycosylation: nature, distribution, enzymatic formation, and disease implications of glycopeptide bonds. Glycobiology, 12(4), 43R-56R.
- Walsh, G., & Jefferis, R. (2006). Post-translational modifications in the context of therapeutic proteins. Nature biotechnology, 24(10), 1241-1252.
- Wijdeven MA, van Geel R, Hoogenboom JH, Verkade JMM, Janssen BMG, Hurkmans I, de Bever L, van Berkel SS, van Delft FL. Enzymatic glycan remodeling-metal free click (GlycoConnect™) provides homogenous antibody-drug conjugates with improved stability and therapeutic index without sequence engineering. MAbs. 2022 Jan-Dec;14(1):2078466. doi: 10.1080/19420862.2022.2078466. PMID: 35634725; PMCID: PMC9154768.



